- 1. What are the different phases of normal wound healing?
- 2. What are the main factors involved in delayed wound healing?
- 3. Which medicinal products affect wound healing?
- 4. What is the impact of topical corticosteroids on wound healing?
- 5. Is there a difference between wound healing in acute and chronic wounds?
- 6. How should you clean wounds?
- 7. What are the specific features of wound healing in children?
- 8. What are the specific features of wound healing in the elderly?
- 9. How to manage wound healing in cancer patients?
- 10. What are the specific features of wound healing in patients with burn injuries?
- 11. What dressing should be used according to the appearance of the wound?
- 12. What role do dermocosmetics play in wound healing?
- 13. Is there a role for alternative therapies in wound healing?
- 14. What advice should be given to patients to achieve an optimal scar (postoperative and post-trauma)?
- 15. What is a pathological scar and why does it develop?
- 16. How do you treat pathological scars?
- 17. How do you treat and heal a leg ulcer?
- 18. How do you treat and heal a pressure sore?
- 19. How do you treat and heal a foot wound in a diabetic patient?
- 20. What is the relationship between wound healing and mental health?
10. What are the specific features of wound healing in patients with burn injuries?
It is estimated that 400,000 people receive a burn injury in France each year (of which more than a third are children). Thus it is important that practitioners are able to accurately assess the wound. This assessment allows the patient to be referred to a specialised unit, if necessary, and be offered a suitable treatment to achieve optimal healing.
The depth of the burns corresponds to the anatomical level affected, and a distinction is made between:
• superficial burns (1st degree and 2nd degree burns): medical treatment and healing without after-effects;
• deep burns (2nd degree and 3rd degree): management generally requires the intervention of a plastic surgeon, and healing will involve after-effects (Figure7 and Table3).
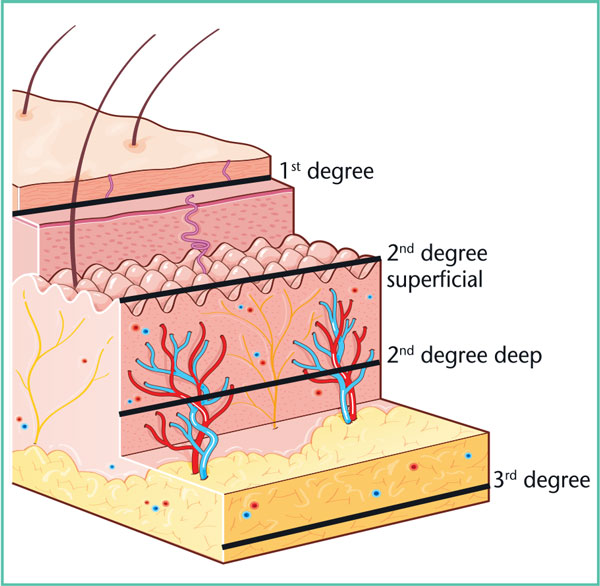
Figure 7. Classification of burns in degrees.
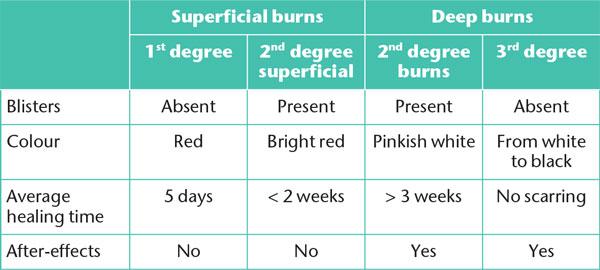
Table 3. Differences in the clinicalvappearance of the burn depending on the degree.
In any burn injury, the wound should first be cooled down under running water for a prolonged time (about ten minutes). Pain management, which is often the primary concern, is essential. In common with all wounds, it is important to check the tetanus vaccination status of the patient.
Superficial burns should be treated by:
• washing with soap and water;
• excision of blisters (except on palmsof the hand and soles of the feet);
• daily tulle-gras dressing.
Healing normally occurs in under two weeks. If this is not the case, the patient should be referred to a specialist centre. Determining the depth of a burn is not always easy and the initial assessment may be incorrect and explain the delay in healing. For this reason, in second-degree burns, a reassessment after 24-48 hours is always required.
Deep burns should always be treated in a specialised burns centre for rapid flattening and grafting.
References
1. Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med 1999;341: 738-46.
2. Eming SA, Krieg T, Davidson JM. Inflammation in wound repair: molecular and cellular mechanisms. J Invest Dermatol2007; 127: 514-25.
3. Suivi en ville des plaies chroniques : ulcère veineux de jambe, escarre, plaie du pied diabétique. Assurance maladie, octobre 2015.
4. Plaies aiguës en structure d’urgence. Référentiel de bonnes pratiques. SFMU, 2017: 32 p.
5. Sgonc R, Gruber J. Age-related aspects of cutaneous wound hea- ling: a mini-review. Gerontology 2013; 59: 159-64.
6. Majtan J. Honey: an immunomodulator in wound healing. Wound Repair Regen 2014; 22: 187-92.
7. Opletalová K, BlaizotX, Mourgeon B, et al. Maggot therapyfor wound debridement: a randomized multicenter trial. Arch Dermatol 2012; 148: 432-8.
8. van den Broek LJ, Limandjaja GC, Niessen FB, Gibbs S. Human hypertrophic and keloid scar models: principles, limitations and future challenges from a tissueengineering perspective. ExpDer- matol 2014; 23: 382-6.
9. Prise en charge de l’ulcère de jambe à prédominance veineuse hors pansement. Recommandations HAS, 2006.
10. La compression médicale dans les affections veineuses chroniques. Fiche de bon usage HAS, 2010.
11. Edmonds M, Lázaro-Martínez JL, Alfayate-García JM, et al. Sucrose octasulfate dressing versus control dressing in patients with neu- roischaemic diabetic foot ulcers (Explorer): an international, multi- centre, double-blind, randomised, controlled trial [published correction appears in Lancet Diabetes Endocrinol 2018]. Lancet Diabetes Endocrinol 2018; 6: 186-96.
12. Reinholz M, Poetschke J, Schwaiger H, Epple A, Ruzicka T, Gauglitz GG. The dermatology life quality index as a means to assess life quality in patients with different scar types. J Eur Acad Dermatol Venereol 2015; 29: 2112-9.

