- 1. What are the different phases of normal wound healing?
- 2. What are the main factors involved in delayed wound healing?
- 3. Which medicinal products affect wound healing?
- 4. What is the impact of topical corticosteroids on wound healing?
- 5. Is there a difference between wound healing in acute and chronic wounds?
- 6. How should you clean wounds?
- 7. What are the specific features of wound healing in children?
- 8. What are the specific features of wound healing in the elderly?
- 9. How to manage wound healing in cancer patients?
- 10. What are the specific features of wound healing in patients with burn injuries?
- 11. What dressing should be used according to the appearance of the wound?
- 12. What role do dermocosmetics play in wound healing?
- 13. Is there a role for alternative therapies in wound healing?
- 14. What advice should be given to patients to achieve an optimal scar (postoperative and post-trauma)?
- 15. What is a pathological scar and why does it develop?
- 16. How do you treat pathological scars?
- 17. How do you treat and heal a leg ulcer?
- 18. How do you treat and heal a pressure sore?
- 19. How do you treat and heal a foot wound in a diabetic patient?
- 20. What is the relationship between wound healing and mental health?
17. How do you treat and heal a leg ulcer?
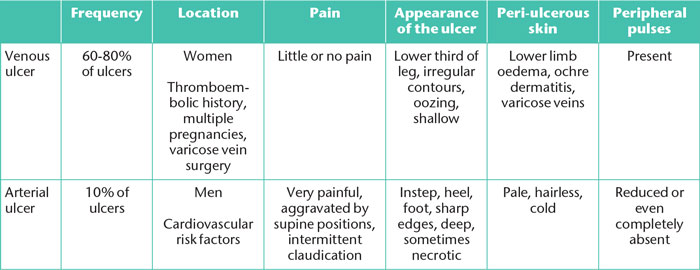
Leg ulcers affect 1% of the general population and 5% of patients over the age of 80 years (HAS 2006). They are most commonly caused by venous insufficiency but can also be caused by arterial problems due to lower limb arteriopathy, or are of mixed origin, combining venous insufficiency and lower limb arteriopathy. Venous ulcers may be the result of venous failure due to leaky valves (leading to varicose ulcers) or may occur after phlebitis, causing deep venous insufficiency triggering venous ulcers that are more difficult to heal.
Treating an ulcer will depend on its origin thus it is essential to determine the nature of the ulcer involved (Table 7). With any lower-limb ulcer, it is advisable to palpate the peripheral pulses, and their absence or reduction indicates a potential ulcer of arterial origin. Another central element is the measurement of the systolic pressure index (SPI). This measurement, calculated using a hand-held Doppler and a manual pressure cuff, is the ratio between ankle systolic pressure/arm systolic pressure. SPI normally ranges between 0.9 and 1.3. When this figure is above 1.3 and the leg arteries cannot be compressed, this is referred to as “mediacalcosis” (i.e. media calcification of arterial vessels). SPI below 0.9 indicates the presence of lower limb arteriopathy, which is considered to be a critical stage of ischaemia if the SPI is below0.5 [9].
If doctors do not have the appropriate equipment in their surgery, they can ask the radiologist to measure the SPI when performing the arterial Doppler ultrasound.
Table 7. Major differences between venous and arterial ulcers.
A vascular assessment is important as:
• in venous ulcers,treatment is based on venous compression [10];
• in arterial ulcers, compression is contraindicated (Figure 12). An arterial Doppler ultrasound of the lower limbs should be performed and the patientreferred to a vascular surgeon.
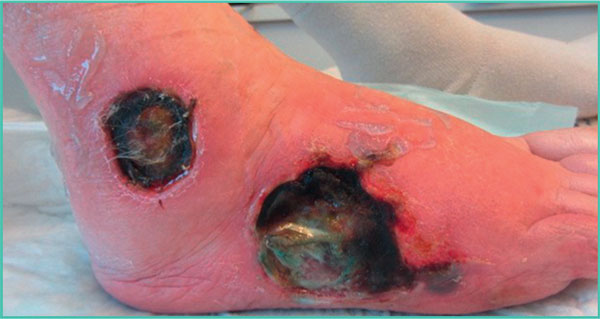
Figure 12. Deep and necrotic arterial ulcers.
References
1. Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med 1999;341: 738-46.
2. Eming SA, Krieg T, Davidson JM. Inflammation in wound repair: molecular and cellular mechanisms. J Invest Dermatol2007; 127: 514-25.
3. Suivi en ville des plaies chroniques : ulcère veineux de jambe, escarre, plaie du pied diabétique. Assurance maladie, octobre 2015.
4. Plaies aiguës en structure d’urgence. Référentiel de bonnes pratiques. SFMU, 2017: 32 p.
5. Sgonc R, Gruber J. Age-related aspects of cutaneous wound hea- ling: a mini-review. Gerontology 2013; 59: 159-64.
6. Majtan J. Honey: an immunomodulator in wound healing. Wound Repair Regen 2014; 22: 187-92.
7. Opletalová K, BlaizotX, Mourgeon B, et al. Maggot therapyfor wound debridement: a randomized multicenter trial. Arch Dermatol 2012; 148: 432-8.
8. van den Broek LJ, Limandjaja GC, Niessen FB, Gibbs S. Human hypertrophic and keloid scar models: principles, limitations and future challenges from a tissueengineering perspective. ExpDer- matol 2014; 23: 382-6.
9. Prise en charge de l’ulcère de jambe à prédominance veineuse hors pansement. Recommandations HAS, 2006.
10. La compression médicale dans les affections veineuses chroniques. Fiche de bon usage HAS, 2010.
11. Edmonds M, Lázaro-Martínez JL, Alfayate-García JM, et al. Sucrose octasulfate dressing versus control dressing in patients with neu- roischaemic diabetic foot ulcers (Explorer): an international, multi- centre, double-blind, randomised, controlled trial [published correction appears in Lancet Diabetes Endocrinol 2018]. Lancet Diabetes Endocrinol 2018; 6: 186-96.
12. Reinholz M, Poetschke J, Schwaiger H, Epple A, Ruzicka T, Gauglitz GG. The dermatology life quality index as a means to assess life quality in patients with different scar types. J Eur Acad Dermatol Venereol 2015; 29: 2112-9.

